 They are generally derived from the source of the compounds. Common names are derived from Greek or Latin words. Many carboxylic compounds are better known by their common name rather than systematic or IUPAC names. Nomenclature of carbonyl and carboxylic compounds Nomenclature of carboxylic compounds The polar nature of the carbonyl group also increases its reactivity.ģ. Reacting species can attack above or below the plane of a carbonyl group. The carbonyl group has a double probability of attachment of reacting species because of its planer shape. Thus, the carbonyl group becomes polar and a nucleophile or an electrophile can attack it. As oxygen is more electronegative than carbon, it attracts the shared pair of electrons more towards itself and becomes partially negative, making carbon partially positive. This is due to the difference in electronegativity between carbon and oxygen. The carbonyl group is a relatively reactive functional group of organic compounds. Reactivity of carbonyl vs carboxyl compounds The fourth orbital of carbon is unhybridized and forms a pi (π) bond with a p orbital of the oxygen atom.Ģ. Three sp 2 hybridized orbital forms three σ bonds, one with oxygen and two with other groups attached to it. Hybridization of carbonyl and carboxylic compoundsīoth carbonyl and carboxylic compounds are sp 2 hybridized. Here is the detailed summary of the parameters of comparison in between carbonyl and carboxylic compounds: 1. Summary of the differences between carbonyl and carboxyl compounds The carboxyl group can be attached with one alkyl group or hydrogen atom because -OH covers one of the sidesĪcetone, formaldehyde, and acetaldehyde, etcĪcetic acid, benzoic acid and formic acid, etc The carbon atom of carbonyl group can be attached to either hydrogen or carbon They are not acidicĬarboxylic compounds can donate a proton (H+) and that is why they are called acidsĬarbonyl groups can be placed anywhere in the hydrocarbon chain except terminalsĬarboxylic acid groups are terminal functional groups When naming carboxylic compounds, a suffix “oic acid” is usedĬarbonyl compounds cannot donate a proton (H+). The suffix “al” or “one” is used when naming carbonyl compounds They have high melting and boiling points They have relatively low melting and boiling points 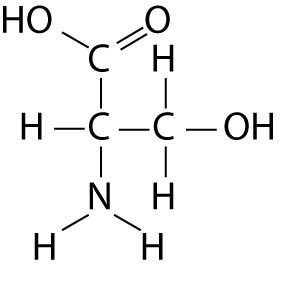
The compounds having COOH functional group are called carboxylic compoundsĬompounds with only carbonyl (C=O) functionality constitute carbonyl compoundsĬompounds with carbonyl (C=O) functional group attached to hydroxyl group constitute carboxylic compoundsĬarbonyl compounds do not have hydrogen bonding with in their moleculesĬarboxylic compounds have strong hydrogen bonding 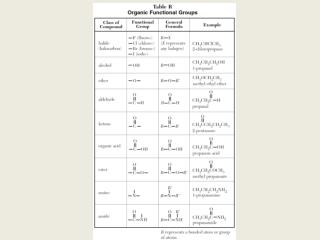
The compounds having C=O functional group are called carbonyl compounds 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
